Water dew point calculator10/13/2023  
Because dew point is a measure of the temperature at which liquid moisture will condense, it is of most benefit in applications where operators need to avoid moisture condensing – for example in pipework where the ambient temperatures may drop to below freezing. It’s stated as ☌ dew point, ☌ frost point or ☌ td The effects of doubling system pressure on dew-point temperatureĮach of these parameters provide operators with a slightly different benefit. As with relative humidity, the dew-point temperature is dependent on the pressure of the gas being measured. Instead of looking at the capacity of the gas to hold moisture, dew point measures the temperature at which liquid moisture will start to condense. It’s stated as parts per million by volume (PPM V)ĭew point is another variable measure of humidity. Unlike relative humidity or dew point, moisture content doesn’t change with pressure or temperature: it’s a measure of the actual molecules of water in the gas. Moisture content is an important parameter for many processes too. The effects temperature has on relative humidity The %rh is a measure of how close to saturation point the humidity in the gas is. If the temperature is lowered or the pressure increase, the saturation point changes and the %rh will also increase. The saturation point varies depending on the temperature of the gas and the pressure with higher temperatures and lower pressures allowing more moisture to be held in the gas as humidity. As with dissolving solids in a liquid, a gas can only hold a certain amount of dissolved moisture before it starts to condense back to liquid phase. Relative humidity is a measure of the humidity in terms of its saturation point. There is always a level of humidity in the air around us, but when it comes to industrial processes, the levels of humidity often need to be controlled. 
Humidity is simply moisture that is dissolved in a gas. What are the differences between humidity, dew point and relative humidity? As well as the term ‘hygrometer’ it may also be called a: Hygrometry is the measurement of the moisture content of gases, and a hygrometer senses, measures and outputs or displays the relative or absolute humidity in the gas. Process moisture is often a contaminant and can severely damage processes, equipment and reduce product quality.It is difficult to measure accurately since moisture is directly affected by temperature and pressure. 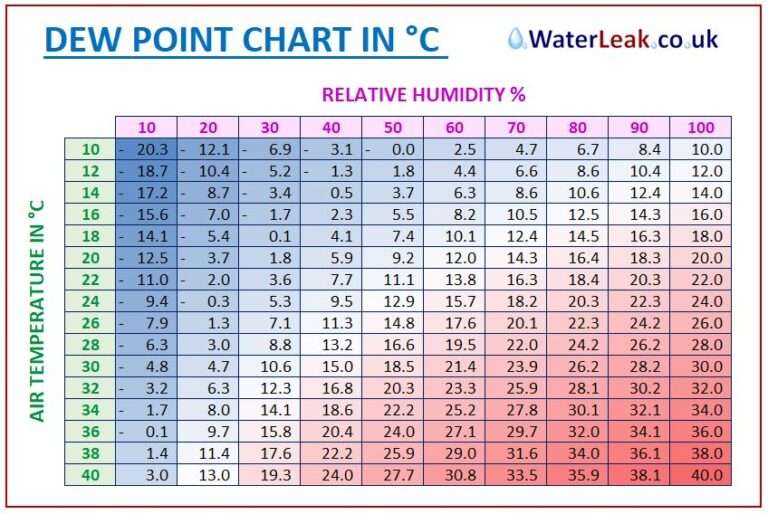
Unlike other process variables such as temperature, pressure, and flow, moisture has two unique characteristics: (If you want to find out about meteorological applications for dew point measurements, here is a short explanation from the UK Met Office.) It’s a common parameter in meteorology, hence the name ‘dew point’ but in this article we are going to look at why it’s important to a wide variety of industrial processes. Where the condensate is ice, this is known as the frost point.’ In that case the measurements would be very inaccurate.The NPL defines dew point as: ‘The temperature at which dew, or condensation, forms as you cool a gas. It is also possible that water may have condensed in the sampling line if it is not properly heated. Dew point tells you what the partial pressure of water vapor is in your sample.ĭid you test the dew point at 8 bar or at ambient? If the sample was tested at ambient then the patial pressure of water in your process is actually 8 times higher than the tested sample. You need to refer to a vapor pressure curve for water. Basically you selected the wrong chart to solve your problem if you are working with H2 gas. The tables that are in weight percent are designed for engineers who often need to calculate weights and percentages. The trick to understanding the humidity tables is to remember its related to the vapor pressure of water and to work (or think)in units of partial pressure (mmHg) as a chemist would. If you add more air to this container and double its pressure the dew point and relative humidity do not change but the weight percent of water in air is cut in half. This air would be called saturated and at 100% relative humidity and the dew point would be 20C. A one liter container of air at 20C cannot contain anymore water vapor than the vapor pressure of water at 20C. In this case it is that water, like any liquid, has a well defined relationship between its temperature and its vapor pressure, and the vapor pressure is not affected by other gasses that are present. can get very complicated if you don't keep in mind the fundamental principles. The topics of relative and absolute humidity, moisture content, dew point, etc.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
